UMBUMB
SINERGISINERGIVoltammetry is widely used to detect heavy metals such as mercury (Hg). The sensor material influences the results of the voltammetry method. Carbon-based sensors are commonly developed because of their wide potential range, low background current, inexpensive, inert, and suitable for various sensors. Recently, Tire Waste (TW) was chosen as a carbon source for the manufacture of electrodes because it is rich in carbon (88%). The material is easy to obtain, green technology and a carbon source that has not been properly utilized. Separation of carbon material from TW using the pyrolysis method produces 4.32 gr (2.6%) of soot (from 200 gr TW) as a material for making carbon-based Hg sensors. The XRD pattern of TW soot has amorphous phases. SEM topography shows that the surface of TW soot consists of particles that are almost uniform in shape. The estimated particle size is about 0.25 µm. The sensor was made with a mixture of TW soot and paraffin with a 2:1 ratio. Testing the specific value of sensor capacitance using the Cyclic Voltammetry (CV) method showed the presence of cathodic and anodic currents. The Hg deposit occurred at a peak cathodic current at a scan rate of 20 mV/s. The Limit of Detection (LoD) value is 0.0681 ppm and the Limit of Quantitation (LoQ) is 0.229 ppm. Measurement of Hg in natural water samples used river water from the Aceh Jaya sub-district at two points representing the upstream and downstream of the river. The Hg concentrations obtained were 0.000536 ppm and 0.00182 ppm, respectively and were compared with results using Atomic Absorbance Spectroscopy (AAS), 0.00058 ppm and 0.00186 ppm, respectively. The t-test results of the two Hg measurement methods at a significance level (α) of 5% obtained tcount > ttable (0.0208 <2.306), indicating that there is a significant difference between the two Hg concentration measurement methods for natural water.
The carbon material (soot) that was successfully separated from 200 grams of TW was 4.Based on the XRD diffractogram, TW soot is estimated to have an amorphous phase.Based on SEM topography the estimated particle size of TW soot is about 250 nm.paraffin for the manufacture of TW carbon-based Hg sensors is 2.The electrochemical response of the UTW carbon electrode-based voltammetry sensor to mercury in water gave LoD and LoQ values of 0.The t-test results of CV with TW carbo-based sensors and AAS for measuring Hg in river water at a significance level (α) of 5% obtained tcount > ttable (6.262), which shows no significant difference between the two concentration measurement methods Hg for river water.
Penelitian lebih lanjut dapat dilakukan untuk mengoptimalkan proses pirolisis limbah ban guna meningkatkan perolehan karbon soot dengan karakteristik yang lebih baik, seperti ukuran partikel yang lebih seragam dan luas permukaan yang lebih besar. Hal ini dapat dicapai melalui variasi parameter pirolisis seperti suhu, waktu tinggal, dan laju pemanasan. Selain itu, eksplorasi penggunaan aktivator selama proses pirolisis, seperti kalium karbonat (K2CO3), dapat meningkatkan porositas dan konduktivitas karbon soot yang dihasilkan, sehingga meningkatkan sensitivitas sensor. Penelitian lanjutan juga perlu difokuskan pada pengembangan sensor berbasis limbah ban yang mampu mendeteksi berbagai jenis logam berat secara simultan, dengan memanfaatkan modifikasi permukaan elektroda menggunakan material selektif seperti nanopartikel logam atau polimer cetak molekul. Pengembangan ini akan menghasilkan sensor yang lebih serbaguna dan efisien untuk pemantauan kualitas air dan lingkungan.
- Solid-State Reference Electrode Based on Thin-Films of Tetrahydrofurfuryl Acrylate (pTHFA) Photopolymer... doi.org/10.1149/2.1331906jesSolid State Reference Electrode Based on Thin Films of Tetrahydrofurfuryl Acrylate pTHFA Photopolymer doi 10 1149 2 1331906jes
- Boron-Doped Diamond Powder as a Durable Support for Platinum-Based Cathode Catalysts in Polymer Electrolyte... doi.org/10.1149/2.0111806jesBoron Doped Diamond Powder as a Durable Support for Platinum Based Cathode Catalysts in Polymer Electrolyte doi 10 1149 2 0111806jes
| File size | 552.1 KB |
| Pages | 8 |
| DMCA | Report |
Related /
ITBITB Penelitian ini memberi bukti bahwa limbah kulit pisang dapat menjadi sumber selulosa berkelanjutan untuk industri kertas, sehingga mengurangi tekanan terhadapPenelitian ini memberi bukti bahwa limbah kulit pisang dapat menjadi sumber selulosa berkelanjutan untuk industri kertas, sehingga mengurangi tekanan terhadap
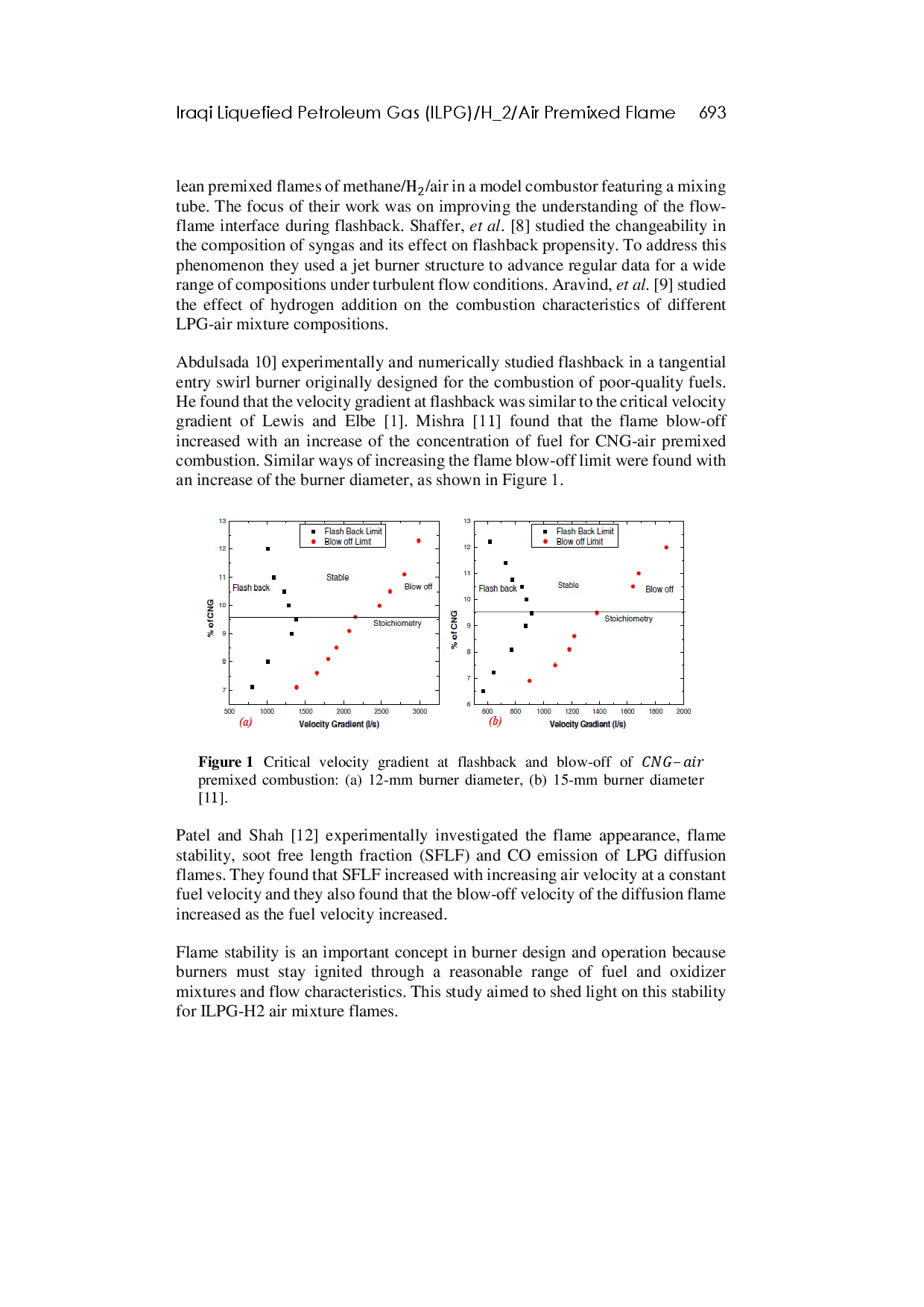
ITBITB Koefisien respons seismik (CS) pada tanah sedang meningkat 10,782% dibanding perhitungan kode saat ini, dan peningkatan lebih tinggi mencapai 13,168% padaKoefisien respons seismik (CS) pada tanah sedang meningkat 10,782% dibanding perhitungan kode saat ini, dan peningkatan lebih tinggi mencapai 13,168% pada
ITBITB Hasil menunjukkan bahwa kertas komposit berbasis kulit pisang memiliki kekuatan tarik dan ketebalan yang memenuhi standar kertas umum, dengan nilai kekuatanHasil menunjukkan bahwa kertas komposit berbasis kulit pisang memiliki kekuatan tarik dan ketebalan yang memenuhi standar kertas umum, dengan nilai kekuatan
ITBITB Jenis penelitian adalah kuasi eksperimen dengan desain pretest–posttest. Populasi adalah seluruh siswa kelas X SMA Negeri 1 Percut Sei Tuan, dan sampelJenis penelitian adalah kuasi eksperimen dengan desain pretest–posttest. Populasi adalah seluruh siswa kelas X SMA Negeri 1 Percut Sei Tuan, dan sampel
ITBITB Abstrak bahasa Indonesia ini menjelaskan bahwa penelitian ini bertujuan untuk mengevaluasi hubungan antara paparan polusi udara dan kejadian penyakit pernapasanAbstrak bahasa Indonesia ini menjelaskan bahwa penelitian ini bertujuan untuk mengevaluasi hubungan antara paparan polusi udara dan kejadian penyakit pernapasan
ITBITB Model inkuiri terbimbing dengan media interaktif secara signifikan meningkatkan kemampuan berpikir kritis dan hasil belajar fisika siswa SMA. PeningkatanModel inkuiri terbimbing dengan media interaktif secara signifikan meningkatkan kemampuan berpikir kritis dan hasil belajar fisika siswa SMA. Peningkatan
ITBITB Menggunakan metode kuasi eksperimen dengan desain pretest-posttest control group, sampel terdiri dari 60 siswa kelas XI di salah satu SMA Negeri di KotaMenggunakan metode kuasi eksperimen dengan desain pretest-posttest control group, sampel terdiri dari 60 siswa kelas XI di salah satu SMA Negeri di Kota
ITBITB Penelitian ini bertujuan melakukan analisis RIc menggunakan kotak reduksi suara (SRB) untuk menilai akurasi dan presisi hasil dibandingkan dengan SIC.Penelitian ini bertujuan melakukan analisis RIc menggunakan kotak reduksi suara (SRB) untuk menilai akurasi dan presisi hasil dibandingkan dengan SIC.
Useful /
STTDBSTTDB Berdasarkan hasil penelitian ergonomi yang dilakukan di area Workshop Grinding PT XYZ, dapat disimpulkan bahwa. Hasil pengukuran antropometri operatorBerdasarkan hasil penelitian ergonomi yang dilakukan di area Workshop Grinding PT XYZ, dapat disimpulkan bahwa. Hasil pengukuran antropometri operator
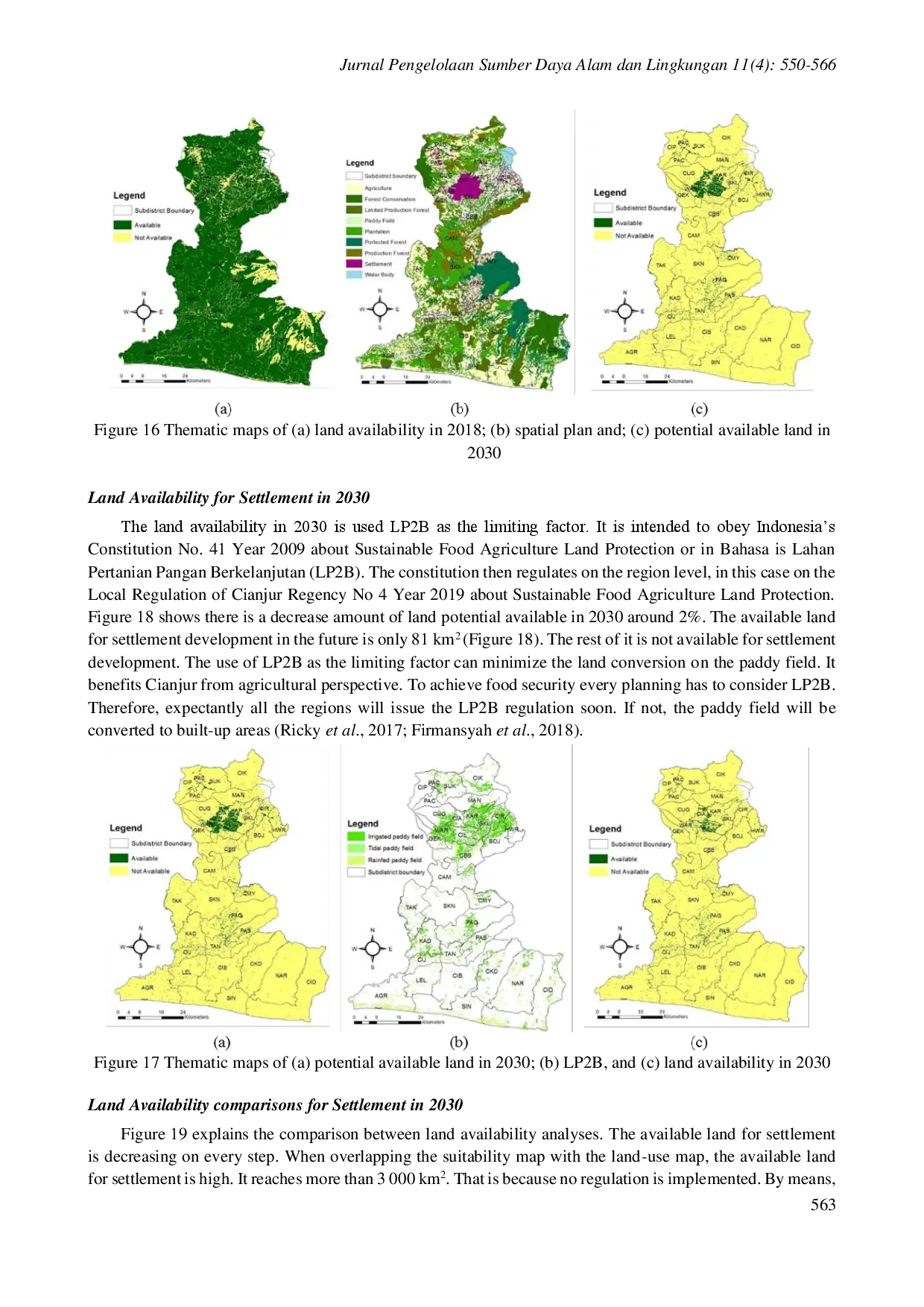
IPBIPB Preferensi pemangku kepentingan terhadap kriteria kesesuaian lahan berbeda, sehingga memengaruhi perencanaan permukiman selanjutnya. Implementasi RencanaPreferensi pemangku kepentingan terhadap kriteria kesesuaian lahan berbeda, sehingga memengaruhi perencanaan permukiman selanjutnya. Implementasi Rencana
UMBUMB Jenis penyimpanan energi mencakup magnet superkonduktor (SMES) dan kapasitif (CES). Jika pengaturan yang benar digunakan, koordinasi PSS, SMES, dan CESJenis penyimpanan energi mencakup magnet superkonduktor (SMES) dan kapasitif (CES). Jika pengaturan yang benar digunakan, koordinasi PSS, SMES, dan CES
UMBUMB Kami menerapkan sistem analogi Newton pada manajemen strategis, kemudian memodelkan dengan turunan formula, diikuti pemodelan simulasi, untuk menentukanKami menerapkan sistem analogi Newton pada manajemen strategis, kemudian memodelkan dengan turunan formula, diikuti pemodelan simulasi, untuk menentukan