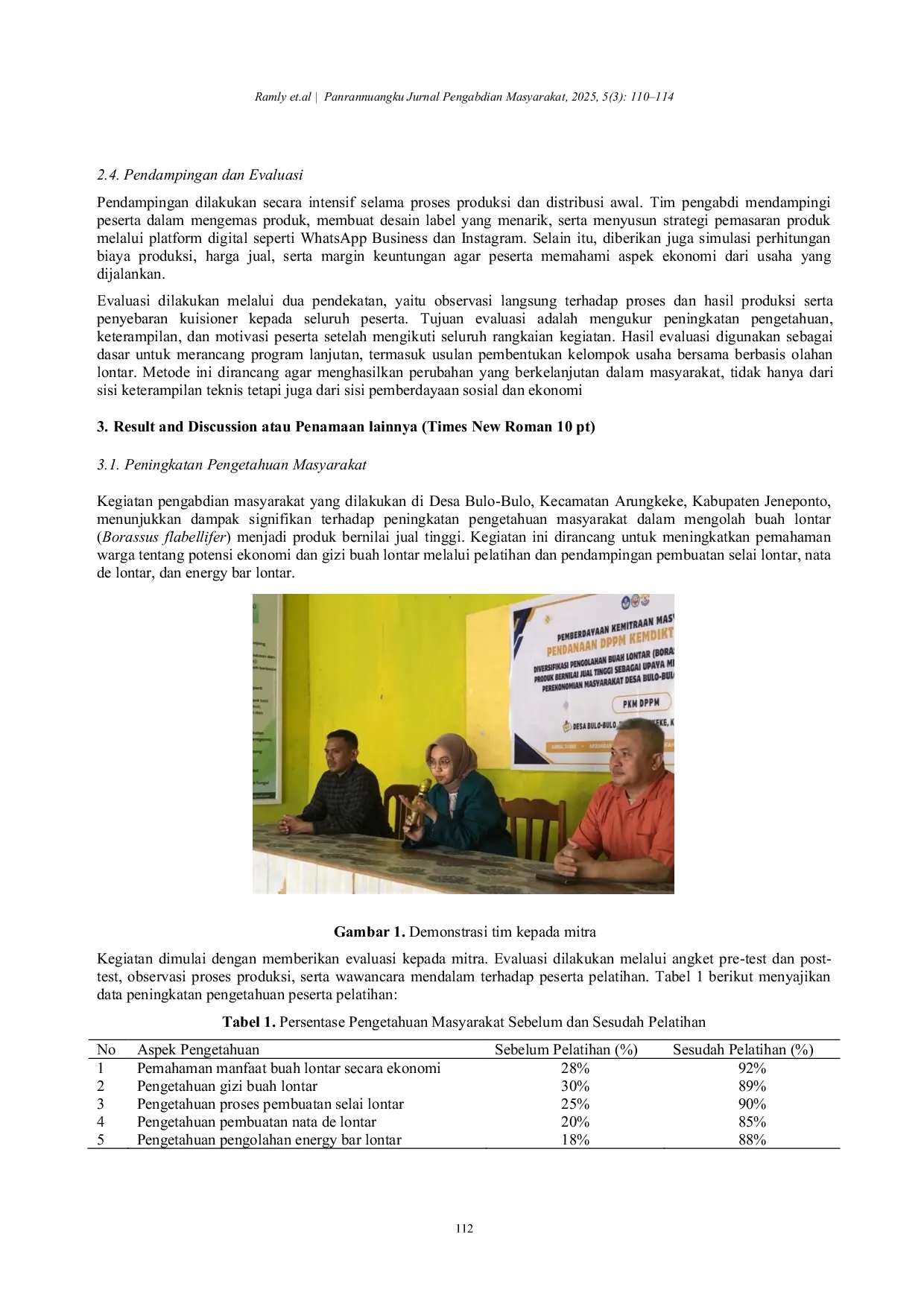
UGMUGM
IJCIJCThis study examines the use of coffee dregs waste as biosorbents of Cu(II) and Ag(I). Coffee dregs waste still contains a high level of carbon and cellulose for biosorbents production. The production process was started with charcoal activation using H3PO4. The batch method was applied by variations of contact time, the mass of the biosorbent, and the initial concentration of metal ions. The results showed that Cu(II) and Ag(I) were optimally adsorbed at pH 6 and 4, respectively. The amount of adsorbed metal ions increased with adsorption contact time. The adsorption process of both metal ions reaches stability within 60 min and the optimum biosorbent mass is 1 g. Isothermal adsorption studies show that Cu(II) adsorption tends to follow Langmuir isotherm with an adsorption energy of 31.42 kJ/mol and Ag(I) adsorption follows Freundlich isotherms with an adsorption energy of 27.74 kJ/mol. Based on the results, the interaction between metal ions and adsorbents is a chemical adsorption process and coffee dregs charcoal has the potential to adsorb Cu(II) and Ag(I) metal ions.
The study demonstrated the potential of H3PO4-activated coffee dregs charcoal as a biosorbent for Cu(II) and Ag(I).Optimal adsorption occurred within 60 minutes, with 1 g of biosorbent proving most effective.The adsorption process for Cu(II) followed the Langmuir isotherm, while Ag(I) followed the Freundlich isotherm, indicating a chemical adsorption mechanism.
Penelitian lebih lanjut perlu dilakukan untuk menguji efektivitas biosorben ampas kopi dalam kondisi lingkungan yang lebih kompleks, seperti keberadaan ion-ion pesaing dan variasi suhu. Studi komparatif dengan jenis biosorben lain, termasuk yang dimodifikasi secara kimia, dapat dilakukan untuk mengidentifikasi keunggulan dan keterbatasan ampas kopi sebagai bahan adsorben. Pengembangan sistem adsorpsi berkelanjutan, misalnya dengan mengintegrasikan biosorben ampas kopi ke dalam sistem filtrasi air limbah skala kecil, dapat menjadi arah penelitian yang menjanjikan untuk aplikasi praktis. Eksplorasi lebih lanjut mengenai mekanisme adsorpsi, termasuk identifikasi gugus fungsi spesifik yang berperan dalam interaksi dengan ion logam, akan memberikan pemahaman yang lebih mendalam tentang kinerja biosorben ini. Penelitian ini diharapkan dapat memberikan solusi inovatif dan ramah lingkungan untuk mengatasi masalah pencemaran logam berat di lingkungan.
| File size | 496.65 KB |
| Pages | 9 |
| DMCA | Report |
Related /
UMGUMG Hal menyebabkannya kurangnya akses mendapatkan oksigen yang bersih dan sehat serta tempat tinggal yang nyaman dan asri bebas dari polusi. Untuk itu ProgramHal menyebabkannya kurangnya akses mendapatkan oksigen yang bersih dan sehat serta tempat tinggal yang nyaman dan asri bebas dari polusi. Untuk itu Program
POLNAMPOLNAM Sistem ini dirancang untuk mengontrol proses pengasapan dengan motor listrik berkecepatan rendah, memungkinkan pengasapan yang konsisten dan efisien. MetodeSistem ini dirancang untuk mengontrol proses pengasapan dengan motor listrik berkecepatan rendah, memungkinkan pengasapan yang konsisten dan efisien. Metode
INSURIPONOROGOINSURIPONOROGO Studi ini juga menggarisbawahi tantangan dalam adaptasi metode pengajaran terhadap kemajuan teknologi pesat serta krusialnya kolaborasi antara pendidik,Studi ini juga menggarisbawahi tantangan dalam adaptasi metode pengajaran terhadap kemajuan teknologi pesat serta krusialnya kolaborasi antara pendidik,
PELITABANGSAPELITABANGSA Hasil penelitian menunjukkan bahwa eceng gondok mampu menurunkan kadar COD, BOD, dan TSS secara signifikan serta menjaga pH tetap dalam kisaran netral.Hasil penelitian menunjukkan bahwa eceng gondok mampu menurunkan kadar COD, BOD, dan TSS secara signifikan serta menjaga pH tetap dalam kisaran netral.
IDJOURNALIDJOURNAL Program ini tidak hanya meningkatkan keterampilan jangka pendek, tetapi juga membentuk ekosistem pembelajaran reflektif yang berkelanjutan. Dengan demikian,Program ini tidak hanya meningkatkan keterampilan jangka pendek, tetapi juga membentuk ekosistem pembelajaran reflektif yang berkelanjutan. Dengan demikian,
IDJOURNALIDJOURNAL Program Pengabdian Masyarakat ini bertujuan untuk memberdayakan Kelompok Tani Ulu Galung 1 di Desa Salassae, Kabupaten Bulukumba melalui penerapan sistemProgram Pengabdian Masyarakat ini bertujuan untuk memberdayakan Kelompok Tani Ulu Galung 1 di Desa Salassae, Kabupaten Bulukumba melalui penerapan sistem
UGMUGM Studi ini menunjukkan bahwa MGONPs adalah adsorben yang lebih baik dengan kapasitas adsorpsi yang lebih tinggi untuk penyerapan BG, dan aplikasinya diStudi ini menunjukkan bahwa MGONPs adalah adsorben yang lebih baik dengan kapasitas adsorpsi yang lebih tinggi untuk penyerapan BG, dan aplikasinya di
UNISAPUNISAP Paparan dalam jangka waktu yang lama menyebabkan stres oksidatif dan perubahan imunologis, termasuk pada sel T yang memproduksi sitokin Interleukin-2 (IL-2).Paparan dalam jangka waktu yang lama menyebabkan stres oksidatif dan perubahan imunologis, termasuk pada sel T yang memproduksi sitokin Interleukin-2 (IL-2).
Useful /
IDJOURNALIDJOURNAL Program ini dilaksanakan bekerja sama dengan MGMP Bahasa Inggris dan melibatkan serangkaian lokakarya berjenjang—pengenalan konsep, praktik merancangProgram ini dilaksanakan bekerja sama dengan MGMP Bahasa Inggris dan melibatkan serangkaian lokakarya berjenjang—pengenalan konsep, praktik merancang
IDJOURNALIDJOURNAL Pengabdian ini meningkatkan literasi digital dan kesadaran masyarakat terhadap pentingnya dokumentasi wisata sebagai dasar pengembangan promosi dan pengelolaanPengabdian ini meningkatkan literasi digital dan kesadaran masyarakat terhadap pentingnya dokumentasi wisata sebagai dasar pengembangan promosi dan pengelolaan
IDJOURNALIDJOURNAL Metode pelaksanaan meliputi sosialisasi potensi buah lontar, pelatihan teknik pengolahan modern, pendampingan pengemasan berbasis food grade, serta pemasaranMetode pelaksanaan meliputi sosialisasi potensi buah lontar, pelatihan teknik pengolahan modern, pendampingan pengemasan berbasis food grade, serta pemasaran
AKPERAKPER Penyakit serangan jantung adalah suatu penyakit yang menyebabkan kematian. Hiperlipidemia adalah sebuah masalah kesehatan masyarakat karena berhubunganPenyakit serangan jantung adalah suatu penyakit yang menyebabkan kematian. Hiperlipidemia adalah sebuah masalah kesehatan masyarakat karena berhubungan