BCRECBCREC
Bulletin of Chemical Reaction Engineering & CatalysisBulletin of Chemical Reaction Engineering & CatalysisHydrogen generation from formic acid using nanoscale zero-valent iron (nZVI) represents a promising route for low-cost and sustainable hydrogen production. However, the effect of sodium borohydride (NaBH₄) molar ratio on nZVI synthesis and performance remains insufficiently explored. This study investigated how varying molar ratios affect nZVI synthesis characteristics and its hydrogen generation efficiency from formic acid, NaBH₄ which acts as a safe and easily handled hydrogen carrier. nZVI was synthesized through a one-step liquid-phase chemical reduction method using NaBH₄ ratios ranging from 4.4:1 to 8.8:1. UV–Vis spectroscopy indicated that the 4.4:1 ratio yielded the highest nZVI formation, reflecting optimal reduction efficiency and particle formation. Hydrogen generation experiments conducted in a closed reactor equipped with a water displacement system revealed that nZVI synthesized at the 4.4:1 ratio achieved the maximum hydrogen volume (98 mL), which progressively declined to 53 mL at the 8.8:1 ratio. These findings demonstrate that precursor molar ratios significantly influence nZVI formation, stability, and reactivity toward hydrogen evolution. An optimal NaBH₄ ratio of 4.4:1 was identified for maximizing nZVI formation and hydrogen volume, providing valuable insights for developing scalable formic acid–based hydrogen generation systems.
This study systematically investigated the effect of NaBH₄ molar ratios on the synthesis of nZVI and its performance in hydrogen generation via formic acid reduction.FeCl₃ molar ratio significantly influenced nZVI formation, with the 4.1 ratio producing the highest absorbance intensity and thus the most effective nZVI formation.Increasing the NaBH₄ proportion led to a gradual decline in absorbance, likely due to the formation of excess boron-rich by-products that promoted particle agglomeration and surface passivation, thereby reducing the amount of reactive nZVI formed.These effects were consistent with the hydrogen generation results, where nZVI synthesized at the 4.1 ratio yielded the highest hydrogen volume (98 mL).FeCl₃ ratios significantly reduced hydrogen output, confirming that precursor molar ratios critically determine the redox activity and reactivity of nZVI in the formic acid reduction pathway.FeCl₃ ratio was found to be optimal for balancing nanoparticle formation, stability, and hydrogen volume.These findings provide valuable insights for designing cost-effective and scalable formic acid-based hydrogen production systems using nZVI.
Berdasarkan penelitian ini, beberapa saran penelitian lanjutan dapat diajukan untuk lebih memahami dan mengoptimalkan produksi hidrogen menggunakan nZVI. Pertama, perlu dilakukan studi kinetika yang lebih mendalam untuk memahami mekanisme reaksi antara nZVI dan asam format pada berbagai rasio molar, sehingga dapat diidentifikasi faktor-faktor yang paling berpengaruh terhadap laju reaksi dan efisiensi produksi hidrogen. Kedua, penelitian dapat difokuskan pada pengembangan material pendukung (support material) untuk nZVI guna mencegah agregasi partikel dan meningkatkan stabilitasnya, serta meningkatkan kemampuan daur ulang nZVI. Ketiga, perlu dilakukan evaluasi kinerja nZVI dalam sistem produksi hidrogen skala kontinu atau pilot, untuk menguji skalabilitas dan keandalan teknologi ini dalam kondisi yang lebih realistis.
| File size | 767.72 KB |
| Pages | 8 |
| DMCA | Report |
Related /
MAHADEWAMAHADEWA Populasi penelitian adalah mahasiswa jurusan pendidikan biologi FPMIPA IKIP PGRI Bali dengan sampel sebanyak 51 mahasiswa. Data dikumpulkan melalui evaluasiPopulasi penelitian adalah mahasiswa jurusan pendidikan biologi FPMIPA IKIP PGRI Bali dengan sampel sebanyak 51 mahasiswa. Data dikumpulkan melalui evaluasi
UNIBUNIB Temuan studi menunjukkan bahwa integrasi pendekatan metode kasus dengan alat teknologi dalam instruksi, silabus, dan materi pelajaran meningkatkan keterlibatanTemuan studi menunjukkan bahwa integrasi pendekatan metode kasus dengan alat teknologi dalam instruksi, silabus, dan materi pelajaran meningkatkan keterlibatan
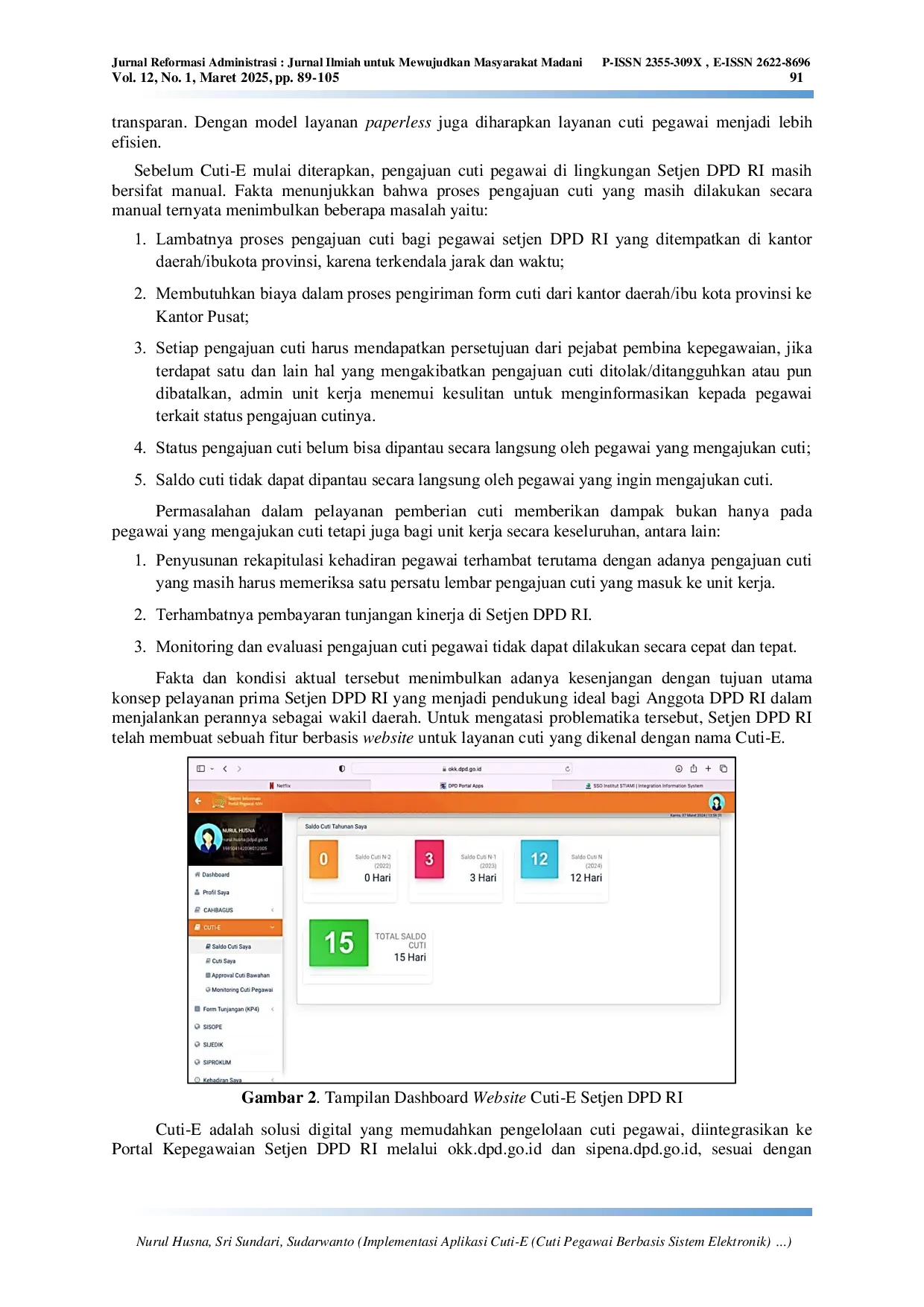
STIAMISTIAMI Keberhasilan Cuti‑E bergantung pada komunikasi yang efektif, manajemen sumber daya manusia, fasilitas digital, serta pengelolaan anggaran yang tepat.Keberhasilan Cuti‑E bergantung pada komunikasi yang efektif, manajemen sumber daya manusia, fasilitas digital, serta pengelolaan anggaran yang tepat.
UADUAD The study confirmed that staying in relationships with intimate partner violence victims is not always due to financial dependence. Fear, especially theThe study confirmed that staying in relationships with intimate partner violence victims is not always due to financial dependence. Fear, especially the
UADUAD Air Traffic Controller (ATC) adalah salah satu profesi di sektor layanan. ATC, seperti pekerjaan lainnya, memiliki tugas sosial lainnya di luar pekerjaanAir Traffic Controller (ATC) adalah salah satu profesi di sektor layanan. ATC, seperti pekerjaan lainnya, memiliki tugas sosial lainnya di luar pekerjaan
MEDIAPUBLIKASIMEDIAPUBLIKASI Hasil Kegiatan telah membawa dampak yang sangat positif bagi para pelaku UMKM di desa tersebut Implementasi website telah berhasil memperluas jangkauanHasil Kegiatan telah membawa dampak yang sangat positif bagi para pelaku UMKM di desa tersebut Implementasi website telah berhasil memperluas jangkauan
UCYUCY Kualitas layanan pendidikan memiliki pengaruh signifikan terhadap kepuasan mahasiswa, yang mengindikasikan pentingnya peningkatan kualitas layanan untukKualitas layanan pendidikan memiliki pengaruh signifikan terhadap kepuasan mahasiswa, yang mengindikasikan pentingnya peningkatan kualitas layanan untuk
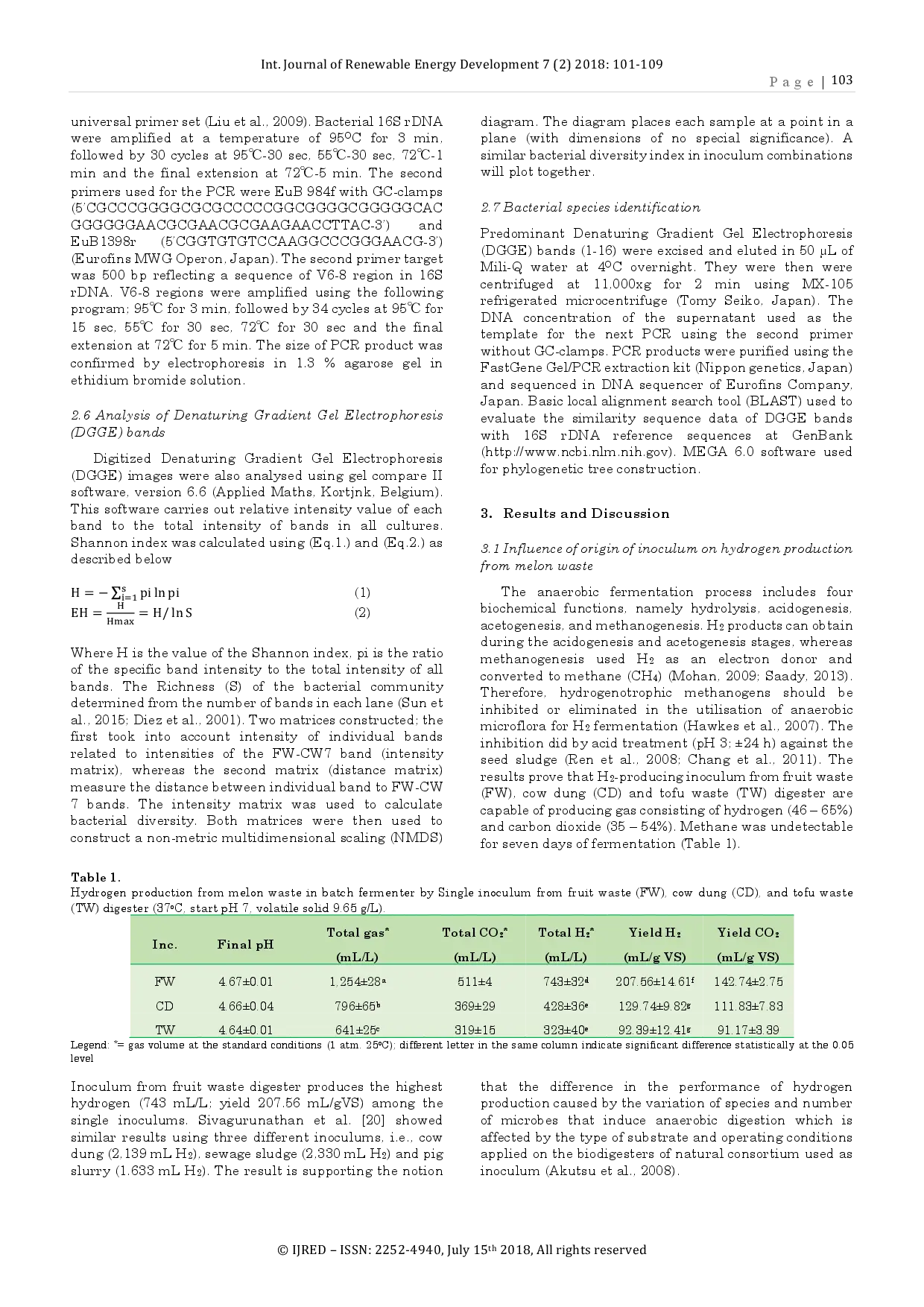
CBIORECBIORE Inokulum dari limbah buah menunjukkan produksi kumulatif hidrogen tertinggi yaitu 743 mL (hasil 207,56 mL/gVS) dan kombinasi inokulum dari limbah buah-kotoranInokulum dari limbah buah menunjukkan produksi kumulatif hidrogen tertinggi yaitu 743 mL (hasil 207,56 mL/gVS) dan kombinasi inokulum dari limbah buah-kotoran
Useful /
MEDIAPUBLIKASIMEDIAPUBLIKASI Kegiatan ini memperkuat sinergi antar UKM melalui berbagi ide dan pengetahuan, serta memberikan manfaat jangka pendek dan potensi dampak positif jangkaKegiatan ini memperkuat sinergi antar UKM melalui berbagi ide dan pengetahuan, serta memberikan manfaat jangka pendek dan potensi dampak positif jangka
MEDIAPUBLIKASIMEDIAPUBLIKASI Dari kegiatan pengabdian kepada masyarakat ini yang dapat disimpulkan bahwa pemahaman ibu-ibu rumah tangga di kelurahan Pantai Amal tentang akuntansi dasarDari kegiatan pengabdian kepada masyarakat ini yang dapat disimpulkan bahwa pemahaman ibu-ibu rumah tangga di kelurahan Pantai Amal tentang akuntansi dasar
CBIORECBIORE Hasil keseluruhan menyimpulkan bahwa briket berkualitas lebih baik dapat diproduksi dari campuran ANH dan SSS dan dapat digunakan untuk berbagai aplikasiHasil keseluruhan menyimpulkan bahwa briket berkualitas lebih baik dapat diproduksi dari campuran ANH dan SSS dan dapat digunakan untuk berbagai aplikasi
CBIORECBIORE The process increases the phenolic content in pyrolysis products, particularly at a 10-minute reaction time. These findings contribute to a better understandingThe process increases the phenolic content in pyrolysis products, particularly at a 10-minute reaction time. These findings contribute to a better understanding